尼羅紅 (染料)
外觀
| 尼羅紅 | |
|---|---|

| |

| |
| IUPAC名 9-(Diethylamino)-5H-benzo[a]phenoxazin-5-one | |
| 別名 | 尼羅藍噁嗪酮 |
| 識別 | |
| CAS號 | 7385-67-3 |
| PubChem | 65182 |
| ChemSpider | 58681 |
| SMILES |
|
| InChI |
|
| InChIKey | VOFUROIFQGPCGE-UHFFFAOYAM |
| ChEBI | 52169 |
| 性質 | |
| 化學式 | C20H18N2O2 |
| 摩爾質量 | 318.376 g/mol g·mol⁻¹ |
| 若非註明,所有數據均出自標準狀態(25 ℃,100 kPa)下。 | |
尼羅紅(英語:Nile red),又稱尼羅藍噁嗪酮(英語:Nile blue oxazone)是一種親脂性染料。可將細胞內的脂滴染成黃色。在大多數極性溶劑中,尼羅紅不會發出熒光;然而在富含脂質的環境中,它會發出強烈的熒光,顏色從深紅色(極性膜脂質)到強烈的黃金色(細胞內儲存的非極性脂質)不等。尼羅紅具有溶劑化顯色特性,其激發波長和發射波長隨溶劑極性變化而變化[1],在極性介質中幾乎不會發出熒光[2]。
尼羅紅在細胞生物學中常作為膜的染色劑,其激發與發射波長與紅色熒光蛋白相同,利用熒光顯微鏡可觀察到其熒光效應。尼羅紅還被用作瓶裝水中微塑料的靈敏檢測所需試劑[3][4]。此外,尼羅紅是製造用於檢測環境變化(如氣味、氣體、pH值等)的不同傳感器膜的候選材料[5] 。
在三酸甘油酯(非極性脂質)中,尼羅紅的最大激發波長約為515 nm(綠色),最大發射波長約為585 nm(黃橙色)[6]。相反,在磷脂(極性脂質)中最大激發波長為554 nm(綠色),最大發射波長為638 nm(紅色)[7]。
合成
[編輯]尼羅紅可以通過尼羅藍與硫酸沸煮水解,尼羅藍中亞胺離子基團被羰基取代轉變成尼羅紅[9]。尼羅紅與其同系物(萘並噁嗪染料)也可以通過5-二烷基氨基-2-亞硝基苯酚與2-萘酚經酸催化縮合得到。此反應不使用共氧化劑,因此產量通常適中[10]。由於生成尼羅紅的反應通常不會完全耗盡尼羅藍,因此則需要額外的分離步驟來獲得純尼羅紅。

-
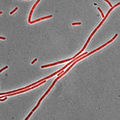
用尼羅紅染色的枯草桿菌。 該菌株部分以細胞鏈的形式生長,因此膜染料可能有助於區分內部細胞邊界
參考文獻
[編輯]- ^ Plenderleith, Richard; Swift, Thomas; Rimmer, Stephen. Highly-branched poly(N-isopropyl acrylamide)s with core–shell morphology below the lower critical solution temperature. RSC Advances. 2014, 4 (92): 50932–50937. doi:10.1039/C4RA10076J. hdl:10454/11180
 .
.
- ^ Greenspan, P; Mayer, E P; Fowler, S D. Nile red: a selective fluorescent stain for intracellular lipid droplets.. The Journal of Cell Biology. 1 March 1985, 100 (3): 965–973. PMC 2113505
 . PMID 3972906. doi:10.1083/jcb.100.3.965.
. PMID 3972906. doi:10.1083/jcb.100.3.965.
- ^ David Shukman. Plastic: WHO launches health review. BBC News Online. 15 March 2018 [2024-07-12]. (原始內容存檔於2024-07-05).
- ^ Mason, Sherri A.; Welch, Victoria G.; Neratko, Joseph. Synthetic Polymer Contamination in Bottled Water. Frontiers in Chemistry. 11 September 2018, 6: 407. Bibcode:2018FrCh....6..407M. PMC 6141690
 . PMID 30255015. doi:10.3389/fchem.2018.00407
. PMID 30255015. doi:10.3389/fchem.2018.00407  .
.
- ^ Khalilian, Alireza; Khan, Md. Rajibur Rahaman; Kang, Shin-Won. Highly sensitive and wide-dynamic-range side-polished fiber-optic taste sensor. Sensors and Actuators B: Chemical. 1 October 2017, 249: 700–707. doi:10.1016/j.snb.2017.04.088.
- ^ Fluorescence SpectraViewer - Nile Red triglycerides. Thermo Fisher Scientific. 2017-05-17 [2020-03-06]. (原始內容存檔於2021-03-16).
- ^ Fluorescence SpectraViewer - Nile Red phospholipids. Thermo Fisher Scientific. 2017-05-17 [2020-03-06].
- ^ Shafiee, Omid; Jenkins, Samantha G.; Ito, Takashi; Higgins, Daniel A. Diffusion of hydrophilic to hydrophobic forms of Nile red in aqueous C12EO10 gels by variable area fluorescence correlation spectroscopy. Physical Chemistry Chemical Physics. 2023-01-27, 25 (4): 2853–2861 [2024-07-12]. ISSN 1463-9084. doi:10.1039/D2CP05578C. (原始內容存檔於2024-07-28) (英語).
- ^ Fowler, S. D.; Greenspan, P. Application of Nile red, a fluorescent hydrophobic probe, for the detection of neutral lipid deposits in tissue sections: comparison with oil red O. Journal of Histochemistry & Cytochemistry. 5 January 2017, 33 (8): 833–836. PMID 4020099. S2CID 10496865. doi:10.1177/33.8.4020099
 .
.
- ^ Park, So-Yeon; Kubota, Yasuhiro; Funabiki, Kazumasa; Shiro, Motoo; Matsui, Masaki. Near-infrared solid-state fluorescent naphthooxazine dyes attached with bulky dibutylamino and perfluoroalkenyloxy groups at 6- and 9-positions. Tetrahedron Letters. 11 March 2009, 50 (10): 1131–1135. doi:10.1016/j.tetlet.2008.12.081.