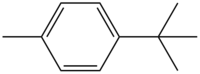
4-甲基叔丁苯
外觀
| 4-甲基叔丁苯 | |
|---|---|
| |
| 別名 | 4-叔丁基甲苯 對甲基叔丁苯 |
| 識別 | |
| CAS號 | 98-51-1 |
| 性質 | |
| 化學式 | C11H16 |
| 摩爾質量 | 148.24 g·mol−1 |
| 密度 | 0.8612 g/cm3(20 °C)[1] |
| 熔點 | −52 °C(221 K)[1] |
| 沸點 | 193 °C(466 K)[1] |
| 若非註明,所有數據均出自標準狀態(25 ℃,100 kPa)下。 | |
4-甲基叔丁苯是一種有機化合物,化學式為C11H16。它可由氫氣在鈀催化下還原4-叔丁基苯甲醛得到[2][3],或在酸催化下由2-甲基丙烯和甲苯反應製得。[4]在相轉移催化劑和氯化鈷催化下,它可以被氧氣氧化為4-叔丁基苯甲酸。[5]它在混酸中硝化,可以得到3-硝基-4-甲基叔丁苯。[6]
參考文獻
[編輯]- ^ 1.0 1.1 1.2 "Hazardous Substances Data Bank" data were obtained from the National Library of Medicine (US). Retrieved from SciFinder. [2021-12-24].
- ^ Shuguo Wang, Peng Zhou, Liang Jiang, Zehui Zhang, Kejian Deng, Yuhua Zhang, Yanxi Zhao, Jinlin Li, Steven Bottle, Huaiyong Zhu. Selective deoxygenation of carbonyl groups at room temperature and atmospheric hydrogen pressure over nitrogen-doped carbon supported Pd catalyst. Journal of Catalysis. 2018-12, 368: 207–216 [2021-12-24]. doi:10.1016/j.jcat.2018.10.017 (英語).
- ^ Giorgio La Sorella, Laura Sperni, Patrizia Canton, Lisa Coletti, Fabrizio Fabris, Giorgio Strukul, Alessandro Scarso. Selective Hydrogenations and Dechlorinations in Water Mediated by Anionic Surfactant-Stabilized Pd Nanoparticles. The Journal of Organic Chemistry. 2018-07-20, 83 (14): 7438–7446 [2021-12-24]. ISSN 0022-3263. doi:10.1021/acs.joc.8b00314. (原始內容存檔於2019-05-02) (英語).
- ^ Fu, Xiangkai; Pu, Wanli; Luo, Bikuei; Deng, Chuanyue. Perfluorooctanesulfonic acid catalyzed Friedel-Crafts alkylation with olefins in gas-liquid phase. Chinese Chemical Letters, 1993. 4 (4): 307-310.
- ^ Nimer Taha, Mandan Chidambaram, Jihad Dakka, Yoel Sasson. Co(II) Catalyzed Solvent Free Auto-Oxidation of Methylbenzenes to Substituted Benzoic Acids Under Phase Transfer Conditions. Catalysis Letters. 2009-05, 129 (3-4): 358–362 [2021-12-24]. ISSN 1011-372X. doi:10.1007/s10562-009-9874-5 (英語).
- ^ J. V. N. Vara Prasad. Synthesis of Heterocyclic Thiosulfonates. Organic Letters. 2000-04-01, 2 (8): 1069–1072 [2021-12-24]. ISSN 1523-7060. doi:10.1021/ol0056170. (原始內容存檔於2021-12-24) (英語).
